From cutting-edge Electron Microscopes to critical Cryo-EM consumables and comprehensive preventative maintenance. We empower research, pharma, and industrial facilities with uncompromising precision.
-
2111, 2nd Cross Rd, D-Block, Gayatrinagar Rajajinagar, Bengaluru, KA 560021
-
Call Us: 97436 20456
(Mon - Sat)

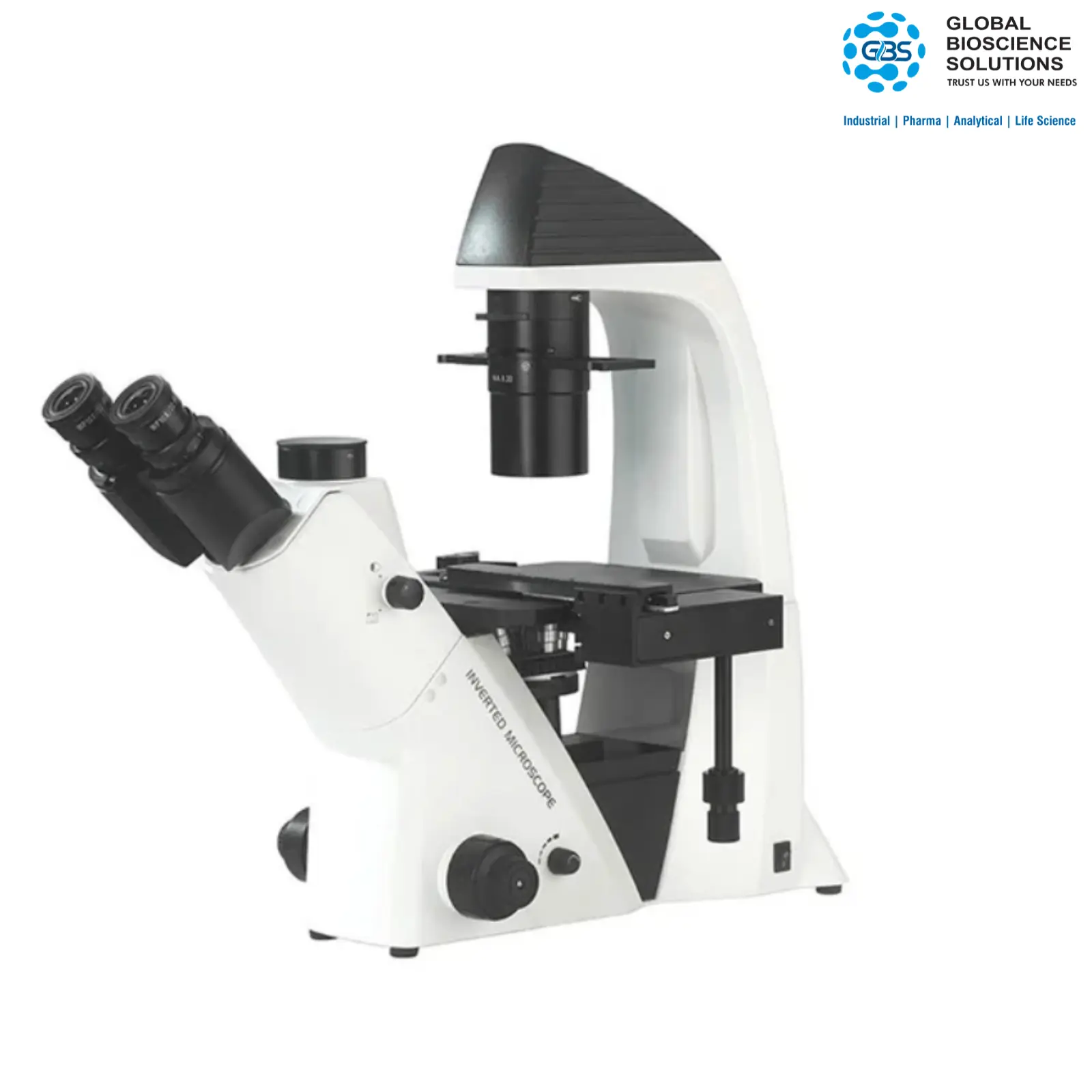
Cosmo-PSA I / II Particle Size Analyzer & Motorized Microscope




Cosmo-PSA I / II Particle Size Analyzer & Motorized Microscope
Editor's Choice: First, the Cosmo-PSA series offers fully automated particle size analysis. Specifically, it pairs easily with a high-precision motorized stage microscope. Therefore, it works perfectly in strict regulatory environments. Most importantly, it features FDA 21 CFR Part 11 compliance. As a result, it is the ideal optical tool for PMT (USP 788/789), API, and Placebo testing.
Includes 1 Year Warranty & On-Site Installation Support
Precision Automated Particle Size Analysis
First, the Cosmo-PSA I and Cosmo-PSA II systems are top-tier automated particle size analyzers. They integrate smoothly with high-performance motorized stage microscopes. Furthermore, these systems use advanced optical detection and a high-resolution CCD camera. As a result, they deliver fast, hands-free sample scanning. In fact, the scanning process takes only about 2 minutes. Therefore, the Cosmo-PSA greatly boosts lab output and reduces human error.
Pharmaceutical & Regulatory Applications
Additionally, the Cosmo-PSA series meets the strict standards of the pharmaceutical industry. Specifically, it excels in PMT (USP 788/789) verification. Moreover, the setup allows thorough testing of API, placebos, and injectables. It can also calculate critical D values easily. Because of this, the motorized stage supports many advanced workflows. For instance, you can use it for 96-well plate scanning, yeast cell counting, and checking chemical crystallinity.
FDA 21 CFR Part 11 Compliant Software
Data security is clearly vital in modern regulated labs. To meet these strict rules, the Cosmo-PSA uses proprietary image analysis software. Most importantly, this software ensures full FDA 21 CFR Part 11 compliance. Consequently, all automated measurements and audit trails remain totally secure. Furthermore, user access histories and electronic signatures are securely locked. Ultimately, this keeps your data fully audit-ready for inspections.
Frequently Asked Questions
1. What are the primary applications of the Cosmo-PSA series?
Primarily, the system is built for strict pharmaceutical quality control. For example, applications include PMT (USP 788/789) and API analysis. Additionally, it tests injectables, counts yeast cells, and determines D values.
2. Is the software compliant with FDA regulations?
Yes, absolutely. The included optical software is fully compliant with FDA 21 CFR Part 11. Therefore, it guarantees secure, traceable, and audit-ready data.
3. How fast is the scanning process?
First, the system uses an automated motorized XY stage. Next, it captures images with a continuous CCD camera. As a result, it completes a standard scan in just 2 minutes.
4. Can the motorized stage accommodate different sample types?
Yes, indeed. The precision mechanical stage is highly versatile. Furthermore, it includes custom holders for standard filter papers and microscope slides. Most importantly, it easily holds standard 96-well plates for high-volume testing.
Request Official Quote
Fast Response: Receive a formal GST invoice and compliance documentation in your inbox within 30 minutes.