
SEM for Pharma QC
in India
How Indian pharma QC labs use scanning electron microscopy for API particle morphology, coating uniformity, tablet analysis, and GMP-compliant quality control — with the Cosmo-PSA for USP 788/789 particle counting.
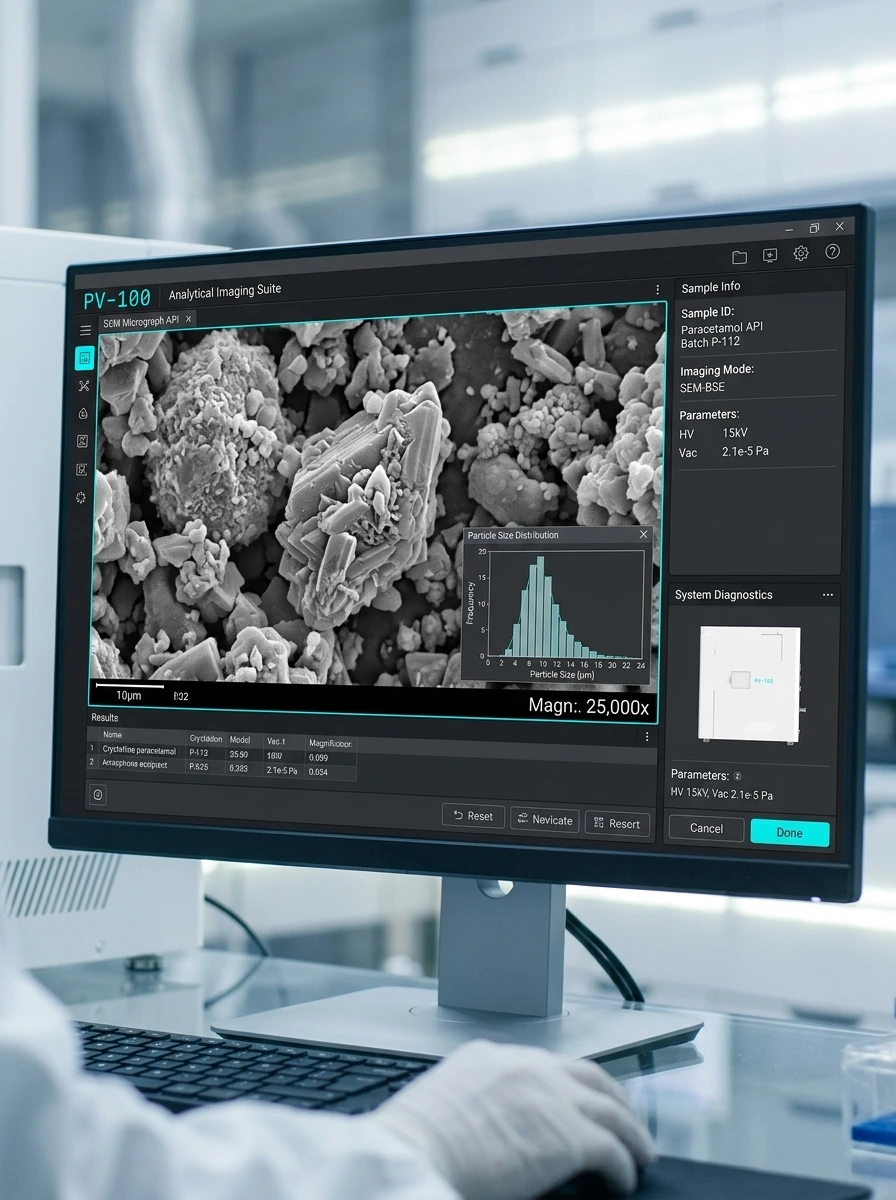
For pharmaceutical QC laboratories in India, scanning electron microscopy (SEM) is the standard technique for API particle morphology, tablet coating uniformity inspection, excipient characterisation, and contamination identification — applications where optical microscopy lacks resolution and where regulatory submissions require morphological data. Global Bioscience Solutions (GBS) supplies the Module Sci PV-100 tabletop SEM (₹60 lakh, 20x–300,000x) and the Cosmo-PSA particle size analyzer (FDA 21 CFR Part 11, USP 788/789 compliant) to Indian pharma companies including Aurigene Pharmaceutical Services, Himalaya Drug Company, and Syngene International — all in Bengaluru.
Why Pharmaceutical QC Labs in India Use SEM
Optical microscopy has served pharmaceutical QC for decades — but it has a hard resolution limit of approximately 200nm. Below this, particle surface features, crystal habit details, coating defects, and nanoscale contamination are invisible. This is where scanning electron microscopy becomes essential.
SEM uses a focused electron beam to scan the sample surface and collect secondary electrons (surface topography) and backscattered electrons (compositional contrast). The result: images at 20x to 300,000x magnification that reveal what no optical system can. For Indian pharma companies filing INDs, NDAs, and ANDAs — or preparing for FDA and WHO GMP audits — SEM provides the particle morphology data that regulators expect to see in physical characterisation packages.

4 Core SEM Applications in Indian Pharma QC
The shape, surface texture, and crystal habit of API particles directly affect dissolution rate, bioavailability, powder flow, and processability. SEM resolves these characteristics at sub-micron level — data that laser diffraction and optical microscopy cannot provide.
- Crystal habit: needle, plate, rod, prismatic, irregular — identified by SEM
- Amorphous vs crystalline surface texture differentiation
- Agglomeration pattern analysis — impact on dissolution and blending
- Post-milling and micronisation verification
- Polymorphic form support alongside XRPD and DSC
Tablet coating quality directly controls drug release profile, stability, and patient safety. SEM cross-sections and surface imaging detect defects that visual inspection and dissolution testing cannot catch early — critical for OOS investigations and formulation development.
- Coating thickness measurement from cross-section SEM imaging
- Pinhole and microcrack detection in enteric coatings
- Delamination and adhesion failure root cause identification
- Multi-layer modified-release coating integrity verification
- Batch-to-batch coating surface uniformity comparison
Surface characteristics of solid dosage forms affect dissolution, stability, and processability. SEM provides direct visual evidence of surface structure — essential for formulation development, stability investigations, and manufacturing failure analysis.
- Tablet surface porosity — impact on wettability and dissolution rate
- Sticking and picking investigation — tablet punch surface interaction
- Lyophilised cake morphology for injectable products
- Spray-dried particle morphology and surface smoothness
- Foreign particle identification and EDS elemental analysis
Excipient quality varies between batches and suppliers. SEM characterises the physical form of incoming materials — a critical incoming QC step that identifies batch-to-batch variation before it reaches the finished drug product.
- MCC — particle morphology and fibre structure batch comparison
- Lactose monohydrate — crystal form and agglomerate detection
- Magnesium stearate — particle size impact on lubrication efficacy
- Starch — granule integrity and particle size range verification
- API-excipient blend uniformity at particle level
SEM in Pharma Regulation — India & FDA
Understanding where SEM fits within the regulatory framework helps pharma QC managers justify the investment and ensures data is used correctly in submissions and audit responses.
| Regulation | Relevance to SEM | SEM Required? | What Applies Instead |
|---|---|---|---|
| ICH Q6A | Recommends physical characterisation including particle morphology for drug substances | Recommended | SEM is the accepted technique for morphology in NDA/ANDA submissions |
| FDA 21 CFR Part 211 | cGMP — requires adequate physical and chemical testing of drug products | As needed | Used for OOS investigations and physical characterisation supporting cGMP records |
| USP <788> / <789> | Particulate matter in injections — requires image-based particle counting | Not SEM | Use Cosmo-PSA (automated image analysis, USP-compliant) — not SEM |
| FDA 21 CFR Part 11 | Electronic records and signatures for regulated data | For data integrity | Cosmo-PSA is fully 21 CFR Part 11 compliant. SEM exports metadata for GMP records. |
| WHO GMP / Schedule M India | Indian GMP requirements including adequate physical characterisation | As needed | SEM satisfies physical characterisation requirements in formulation development and QC investigation workflows |
| EMA CTD Module 3 | Quality dossier — physical characterisation of drug substance and product | Recommended | SEM morphological data supports CTD Module 3 for companies filing in Europe |
SEM Workflow for Pharma QC — Step by Step
The following is the standard SEM workflow in Indian pharma QC labs using the Module Sci PV-100 from GBS. Total time from sample to image: approximately 30–45 minutes per sample set.
Sample Preparation — Mount on SEM Stub
Affix a carbon adhesive tab to a 12.5mm or 25mm aluminium SEM stub. Disperse a small quantity of the pharma sample (API powder, tablet fragment, excipient) onto the tab. For tablet coatings, use a cross-section in epoxy or mount the coated surface directly.
Time: ~5 minutesSputter Coating — Apply Conductive Layer
Pharma samples are almost universally non-conductive and require gold or carbon sputter coating before SEM imaging. Gold (3–5 nm) for imaging only. Carbon (5–8 nm) when EDS elemental analysis is also needed. GBS supplies Quorum sputter coaters alongside the PV-100.
Document coating parameters in batch record for GMP complianceSample Loading — Sub-60 Second Exchange
Load the stub into the PV-100 motorised 5-axis stage. The PV-100’s sub-60-second sample exchange enables rapid multi-sample sessions — critical for QC labs running multiple API or excipient batches in a single session.
Sample exchange: <60 secondsOverview Imaging — Survey & Select ROI
Begin at 200x–500x to survey the sample population and identify representative regions of interest (ROI). Use motorised X-Y navigation to systematically scan multiple areas for a statistically representative set of images.
Magnification: 200x–2,000x for overviewHigh-Resolution Characterisation Imaging
Increase magnification to characterise the specific feature: crystal habit (5,000x–50,000x), coating defects (2,000x–10,000x), surface texture (10,000x–50,000x). Optimise accelerating voltage for sample type — lower kV for beam-sensitive biological or polymer samples.
Magnification: 2,000x–50,000x for pharma characterisationEDS Analysis (if Contamination Identification Required)
If a foreign particle or unknown coating composition requires elemental identification, run EDS point analysis or elemental map. Requires carbon-coated samples — not gold. Compatible EDS detectors: Oxford Instruments and Bruker (both available from GBS).
EDS: Oxford or Bruker detector — available from GBSData Export & GMP Documentation
Export images with scale bars, magnification, accelerating voltage, operator, date, and time metadata. For GMP records: document instrument ID, sample ID, preparation method, coating details, and image file names. Module Sci software exports with embedded metadata satisfying ALCOA data integrity requirements.
GMP ALCOA data integrity requirement

SEM vs Cosmo-PSA — Two Instruments, Two Different Questions
These instruments are complementary — not alternatives. Most Indian pharma QC labs that buy a SEM from GBS also purchase the Cosmo-PSA. Here is exactly how to determine which instrument each application requires.
- ✓ API crystal habit and surface texture
- ✓ Coating defect identification (pinholes, delamination)
- ✓ Foreign particle visual identification
- ✓ Excipient morphology comparison
- ✓ OOS investigation — root cause imaging
- ✓ EDS elemental identification of unknowns
- ✓ Tablet cross-section coating thickness
- ✗ USP 788/789 compliant particle counting
- ✗ FDA 21 CFR Part 11 certified data
- ✗ Statistical particle size distribution
- ✓ USP 788 and USP 789 compliant counting
- ✓ FDA 21 CFR Part 11 — full audit trail
- ✓ Statistical particle size D10, D50, D90
- ✓ Automated counting of thousands of particles
- ✓ Injectable QC — particulate matter limits
- ✓ 96-well plate automation
- ✓ IQ/OQ/PQ validation support from GBS
- ✗ Nanoscale surface morphology
- ✗ Crystal habit characterisation
- ✗ EDS elemental analysis
Cosmo-PSA — For Regulatory-Compliant Particle Counting
One of the very few instruments in India with complete USP 788/789 and FDA 21 CFR Part 11 compliance in a single locally-supported system. Supplied by GBS from Bengaluru.
- FDA 21 CFR Part 11 — electronic audit trail, access controls, e-signatures
- USP 788 — particles ≥10µm and ≥25µm in injectable preparations
- USP 789 — particulate matter in ophthalmic solutions
- Image-based automated counting — USP-required microscopic method
- Applications: API, Placebo, Injectables, D-values, 96-well plate, Yeast cells
- IQ/OQ/PQ validation documentation from GBS

Indian Pharma Companies Using GBS Instruments
All three customers below are headquartered in Bengaluru — the same city as GBS — enabling same-day instrument support and close collaboration with QC and R&D teams.
Leading Indian CRO providing drug discovery and development services to global pharma clients. Analytical labs use GBS instruments for FDA-ready characterisation data supporting IND and NDA packages.
One of India’s most trusted pharma and wellness brands. QC labs use GBS instruments for raw material testing and physical characterisation of both synthetic and herbal API particles — where morphology varies between botanical batches.
Publicly listed CRO/CMO providing integrated discovery, development, and manufacturing services. GMP-compliant facilities demand instruments with full audit trail and data integrity — matching GBS’s compliance-capable range.
Setting Up a GMP-Compliant SEM Lab in India
For Indian pharma companies using SEM within or adjacent to GMP manufacturing, the following table maps documentation requirements to GBS support.
| GMP Requirement | What’s Expected | GBS Support |
|---|---|---|
| Instrument Qualification | IQ (Installation), OQ (Operational), PQ (Performance) documentation | ✓ IQ report at commissioning. OQ/PQ support docs on request. |
| Calibration Records | Regular calibration with traceable standards; calibration certificate | ✓ Calibration verification and certificate at each annual AMC PM visit |
| Operator Training Records | Evidence that operators are trained on the specific instrument | ✓ Training certificate provided at installation visit by GBS engineer |
| Data Integrity (ALCOA) | Attributable, Legible, Contemporaneous, Original, Accurate records | ✓ Module Sci software exports images with embedded metadata (operator, date, time, kV, magnification) |
| Preventive Maintenance Schedule | Written PM schedule; PM records maintained in instrument log | ✓ GBS AMC plan = written PM schedule. PM report issued after each annual visit. |
| SOP Framework | Written SOPs for operation, sample preparation, data handling | Advisory GBS provides application notes as SOP framework. Customer QA team finalises. |
Frequently Asked Questions
SEM is used in Indian pharma QC labs for four core applications: API particle morphology (crystal habit, surface texture), tablet coating uniformity (pinholes, delamination, thickness), excipient characterisation (incoming QC, batch comparison), and contamination identification (foreign particle location + EDS elemental mapping). Aurigene Pharmaceutical Services, Himalaya Drug Company, and Syngene International — all in Bengaluru — use GBS-supplied instruments for these applications.
SEM is not explicitly mandatory but is widely used to meet regulatory expectations. Under ICH Q6A, particle morphology characterisation is recommended for drug substance submissions. Under FDA 21 CFR Part 211, manufacturers must adequately characterise products — SEM provides the morphological data. For injectable QC under USP 788/789, use the Cosmo-PSA (automated counting) — not SEM. Both instruments address different compliance needs and most labs need both.
SEM provides high-resolution morphological images — crystal habit, surface texture, coating quality, individual particle investigation. The Cosmo-PSA performs regulatory-compliant statistical particle counting — USP 788/789, FDA 21 CFR Part 11, full audit trail, automated across thousands of particles. These are complementary instruments. GBS supplies both from Bengaluru — contact us for bundle pricing.
API particle overview: 200x–2,000x. Crystal habit and surface texture: 5,000x–50,000x. Coating defect inspection: 500x–5,000x. Contamination particle identification: 20,000x–100,000x. The Module Sci PV-100 SEM covers 20x to 300,000x — the full range of pharma QC imaging requirements in one tabletop instrument.
Yes. Pharma samples (API powders, tablet coatings, excipients) are non-conductive and require sputter coating before SEM imaging. Gold coating (3–5 nm) for imaging only. Carbon coating (5–8 nm) when EDS is also needed. Coating takes 60–120 seconds. GBS supplies Quorum sputter coaters alongside the PV-100 — both from the same supplier, same AMC.
The Module Sci PV-100 is priced at ₹60 lakh. Full-size SEM systems cost ₹80 lakh to ₹2 crore+ and require dedicated rooms. The PV-100 provides equivalent capability for all standard pharma QC applications — API morphology, coating inspection, contamination ID — at significantly lower total cost of ownership. Contact GBS at +91 97436 20456 for pharma bundle pricing with Cosmo-PSA.
GBS supplies scientific instruments to Aurigene Pharmaceutical Services, Himalaya Drug Company, and Syngene International — all in Bengaluru. GBS headquarters is also in Bengaluru at Rajaji Nagar, enabling rapid delivery and same-day support. Call +91 97436 20456 or email sales@globalbiosciencesolution.com.
21 CFR Part 11 applies to electronic records and signatures, not to instruments directly. The SEM software must support audit trail and metadata if SEM data is submitted to FDA. The Module Sci PV-100 exports images with embedded metadata. For particle counting data requiring full 21 CFR Part 11 compliance (injectable QC), the Cosmo-PSA from GBS is purpose-built with complete compliance.
References & External Sources
- [1] ICH Q6A — Specifications: Test Procedures for New Drug Substances and Products — ema.europa.eu
- [2] FDA 21 CFR Part 211 — Current GMP for Finished Pharmaceuticals — ecfr.gov
- [3] USP <788> Particulate Matter in Injections — usp.org
- [4] FDA 21 CFR Part 11 — Electronic Records; Electronic Signatures — fda.gov
- [5] GBS Module Sci PV-100 SEM Specifications — globalbiosciencesolution.com/products/pv-100-sem-specifications
- [6] GBS Pharma Lab Equipment India — globalbiosciencesolution.com/applications/pharma-lab-equipment-india
- [7] GBS Tabletop SEM Buyer’s Guide India 2026 — globalbiosciencesolution.com/guides/tabletop-sem-microscope-india-2026
- [8] GBS Brand Facts — globalbiosciencesolution.com/brand-facts
Set Up Your Pharma QC SEM Lab with GBS
GBS supplies the Module Sci PV-100 SEM and Cosmo-PSA to Indian pharma companies from Bengaluru. Same-day installation, IQ/OQ documentation, and AMC from day one.

